Eva Clemente (ESR 7) from the Monopoli Group at the Royal College for Surgeons in Ireland (RCSI) has co-authored a new publication titled “Probing the glycans accessibility in the Nanoparticle biomolecular corona” and published in Elsevier’s Journal of Colloid and Interface Science.
This paper results from an international collaboration with researchers at CIC biomaGUNE and the company Ludger Ltd., two beneficiaries in the NanoCarb project.
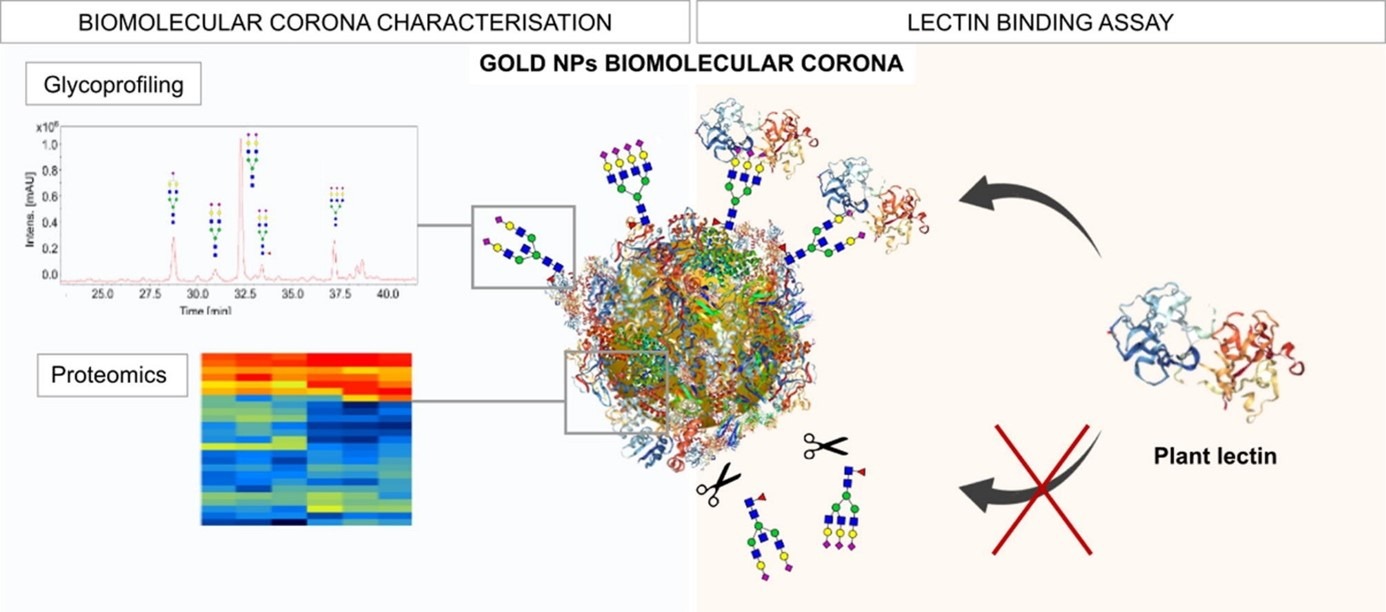
In this paper, 50 nm gold spheres were used to deeply characterize the biomolecular corona obtained at two different plasma concentrations, 10% and 80% v/v. Thus, a lectin binding assay was carried out to assess whether proteins with known affinity towards specific glycans would bind to the corona.
Proteomics and glycol-profiling analysis highlighted that the protein corona composition is dependent on the exposing conditions, while the glyco-profiling is overall very similar, with a net prevalence of sialylated biantennary glycans.
Two plant lectins were employed, Sambucus Nigra (SNA) and Wheat Germ Agglutinin (WGA), both with a high affinity towards terminal sialic acids. Their interaction with the two systems proves the biological accessibility of the glycans on the proteins of the corona. The enzymatic removal of these glycans causes a sensitive loss of binding, proving the fundamental role of the glycans in such interactions.
This study shows for the first time that the glycans are present in the corona and that they could potentially be responsible for the modulation of NP biological processes as they can directly engage with glycan-binding receptors that are highly expressed in an organism.
The paper is available Open Access on the Journal’s website.